 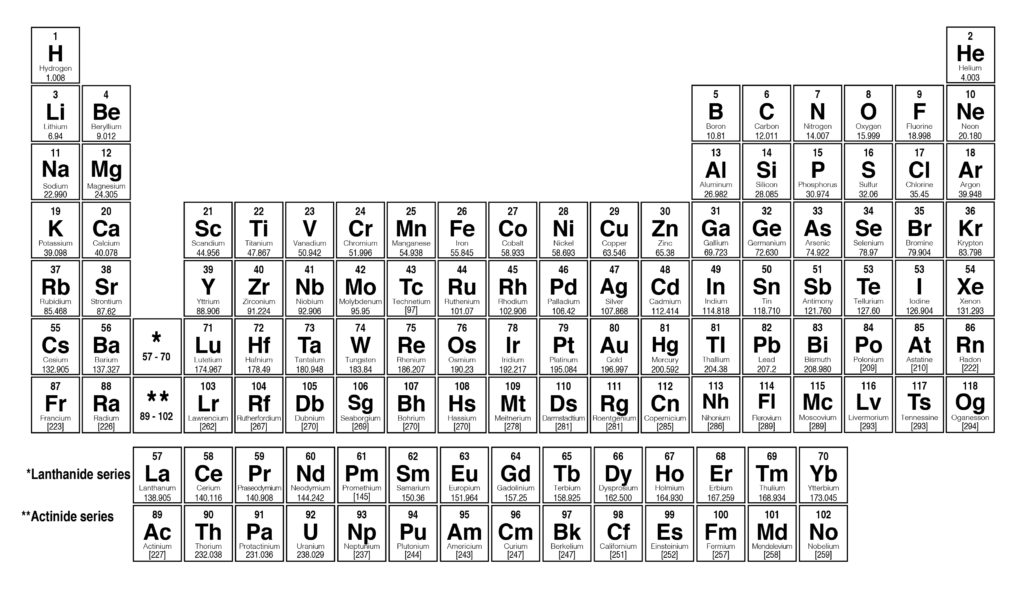
In science, simplicity and beauty are often equated - and that makes hydrogen as beautiful as they come, a single proton and a lone electron making the most compact element in existence. Notice how this approach has trouble with the transition metals. Forget 10 Downing Street or 1600 Pennsylvania Avenue, the most prestigious address in the universe is number one in the periodic table, hydrogen. 
In Group VII, one atom of fluorine (F), chlorine (Cl), and bromine (Br), reacts with one atom of hydrogen. For example, in Group I, two atoms of hydrogen (H), lithium (Li), sodium (Na), and potassium (K) form compounds with one atom of oxygen. The groups in Mendeleev's table are determined by how many oxygen or hydrogen atoms are needed to form compounds with each element. Atomic masses are shown after equal signs and increase across each row from left to right. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. In Meyer’s plot of atomic volume versus atomic mass, the nonmetals occur on the rising portion of the graph, and metals occur at the peaks, in the valleys, and on the down slopes.įigure 3.4.3 Mendeleev’s Periodic Table, as Published in the German Journal Annalen der Chemie und Pharmacie in 1872: The column headings “Reihen” and “Gruppe” are German for “row” and “group.” Formulas indicate the type of compounds formed by each group, with “R” standing for “any element” and superscripts used where we now use subscripts. Bromine is a chemical element with atomic number 35 which means there are 35 protons in its nucleus. \)Īs shown in Figure 3.4.2, the alkali metals have the highest molar volumes of the solid elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
